|
The advantages of gradient gels over fixed-percentage gels are that samples containing a large range of molecular weights can be separated, and proteins with very similar molecular weights can be resolved. This means that at the top of the gel there is a large pore size (e.g., 5% acrylamide) but as the sample moves down through the gel, the pore size decreases (e.g., 20%). 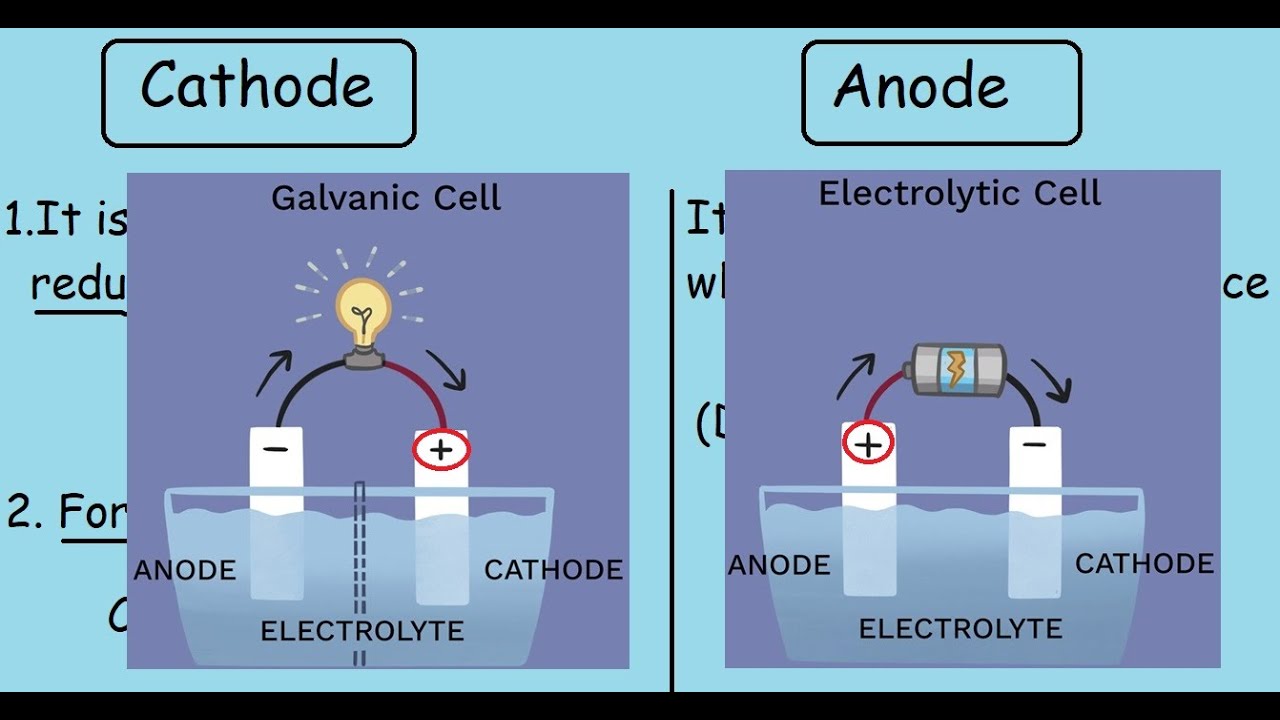
These are gels in which the acrylamide concentration varies uniformly from the top to the bottom of the gel, typically 5% at the top to 25% at the bottom. Gels of between 10 and 20% acrylamide are typically used in SDS-PAGE, where the pore size is small enough to introduce a sieving effect that contributes to the separation of proteins according to their size. The pore size in the gel can be varied by changing the concentrations of both acrylamide and bis-acrylamide used to make the gel, with larger pore sizes resulting from lower percent acrylamide. A porous gel may act as a sieve by retarding, or in some cases completely obstructing, the movement of large macromolecules while allowing smaller molecules to migrate freely. The support matrix provides a means of separating molecules by size, in that they are porous gels. The gel is immersed within an electrophoresis buffer that provides ions to carry a current and some type of buffer to maintain the pH at a relatively constant value. Normally, the gel is cast in the shape of a thin slab, with wells for loading the sample. Protein electrophoresis takes place within a support matrix or "gel", the most commonly used one being polyacrylamide. In denaturing SDS-PAGE separations therefore, migration is determined not by intrinsic electrical charge of the polypeptide, but by molecular weight. It binds to proteins fairly specifically in a mass ratio of 1.4:1, giving a negative charge to the polypeptide in proportion to its length. SDS is an anionic detergent which denatures proteins by "wrapping around" the polypeptide backbone. β-mercaptoethanol or dithiothreitol reduce the disulfide bridges that hold the proteins’ tertiary structure, and the SDS binds strongly to, and denatures, the protein. 
Samples to be run on SDS-PAGE are first boiled in sample buffer containing β-mercaptoethanol or dithiothreitol and SDS. The most widely used method for analyzing protein mixtures qualitatively is SDS-PAGE, which stands for sodium dodecyl sulfate (SDS) polyacrylamide gel electrophoresis (PAGE). At a given pH therefore, and under non-denaturing conditions (the proteins are in their native state), the electrophoretic separation of proteins is determined by both size and charge of the molecules. That is, the charge carried per unit mass of molecule is different from one protein to the other. In addition, the net charge carried by a protein is independent of its size. Below its isoelectric point, the protein is positively charged and migrates towards the cathode. At pH above its isoelectric point, a protein has a net negative charge and migrates towards the anode in an electrical field. Their net charge is determined by the pH of their environment. As such, electrophoresis is one of the most widely-used techniques in biochemistry and molecular biology. Since many important biological molecules, such as nucleic acids and proteins, have ionizable groups, they exist as electrically charged species at any given pH. As an analytical tool, electrophoresis is simple, rapid and highly sensitive. Their rate of migration depends on the strength of the field, on the net charge, size and shape of the particles (i.e., molecules) and also on the ionic strength, viscosity and temperature of the medium in which the molecules are moving. Positively charged particles migrate towards the cathode, and the negatively charged ones towards the anode. Electrophoresis is the migration of a charged particle under the influence of an electric field.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
